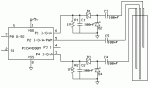
Hi,
I want to detect the level of liquid in a tube.
It is only about 8mm diameter, and contains 95degree C liquid. It is about 5% ethanol in water.
I used simple soldered barfe wires, but found that they corrode very quickly. I then made the electrodes from silver solder, but found they formed a compound between them very quickly.
First question is can I reduce the current enough so that there is not so many electolytic actions and still measure the sensors? If not, I will do it another way. Would stainless steel probes solve the problem?
My other question is, if you CAN use the above method, what is the simple circuit for providing different ADC values for 2 different probes, plus the condition of having no liquid covering either of them?
I made a voltage divider and added another series resisitor to one of the sets of probes, but could not get very good differentiation in values. Can you give me an idea of the circuit and values that should be used?
I want to detect the level of liquid in a tube.
It is only about 8mm diameter, and contains 95degree C liquid. It is about 5% ethanol in water.
I used simple soldered barfe wires, but found that they corrode very quickly. I then made the electrodes from silver solder, but found they formed a compound between them very quickly.
First question is can I reduce the current enough so that there is not so many electolytic actions and still measure the sensors? If not, I will do it another way. Would stainless steel probes solve the problem?
My other question is, if you CAN use the above method, what is the simple circuit for providing different ADC values for 2 different probes, plus the condition of having no liquid covering either of them?
I made a voltage divider and added another series resisitor to one of the sets of probes, but could not get very good differentiation in values. Can you give me an idea of the circuit and values that should be used?